Enovis Foot & Ankle Devices
Foot & Ankle
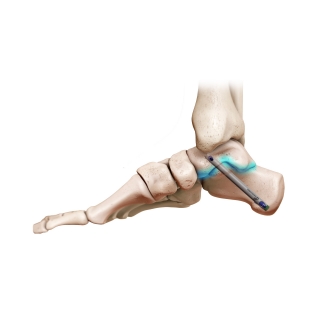
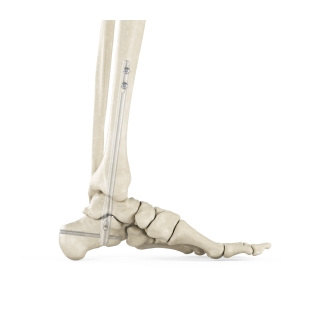
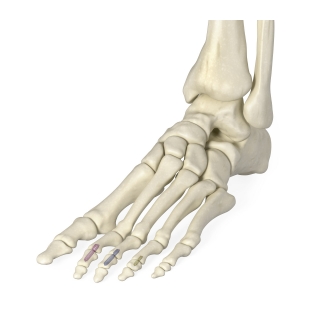
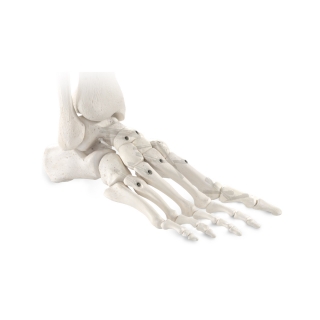
Enovis®, one of the leading medical device companies, is committed to partnering with surgeons to bring innovative technologies to the market and elevating the options available to treat patients effectively. Our products cover a wide range of foot and ankle solutions, from total ankle replacement and innovative static fixation to dynamic compression and fusion. Our mission is to bring differing technologies that are backed by science to the market. Our goal as one of the leading medical device companies for foot and ankle is to address and offer patient options through the continuum of care.
Digital Product Catalog
Interactive catalog of Enovis' F&A portfolio.
IFUs
Instructions for Use.